
Colorado FDA Newswire
-
Published on 06:00 GMT
 Source: Food Safety News - Neutral
Source: Food Safety News - Neutral -
Published on Mar 8, 2026

Coconut Water Recall Update As FDA Sets Risk Level
Around 12,000 cases of coconut water, which were originally voluntarily recalled by Tipp Distributors Inc—a USA-based food and beverage importer—on February 25, 2026, were given a Class III (low) risk level by the U.S. Food and Drug Administration (FDA) on …
Source: Newsweek - Left-wing -
Published on 09:42 GMT

Newsom Picks a Dogfight With Trump and RFK Jr. on Public Health
SACRAMENTO, Calif. — California Gov. Gavin Newsom has positioned himself as a national public health leader by staking out science-backed policies in contrast with the Trump administration. After Health and Human Services Secretary Robert F. Kennedy Jr. …
Source: KHN - Neutral -
Published on 10:24 GMT

Newsom's fight with Trump and RFK Jr. on public health
SACRAMENTO — California Gov. Gavin Newsom has positioned himself as a national public health leader by staking out science-backed policies in contrast with the Trump administration. After Health and Human Services Secretary Robert F. Kennedy Jr. fired …
Source: Los Angeles Times - Center-left -
Published on 09:13 GMT
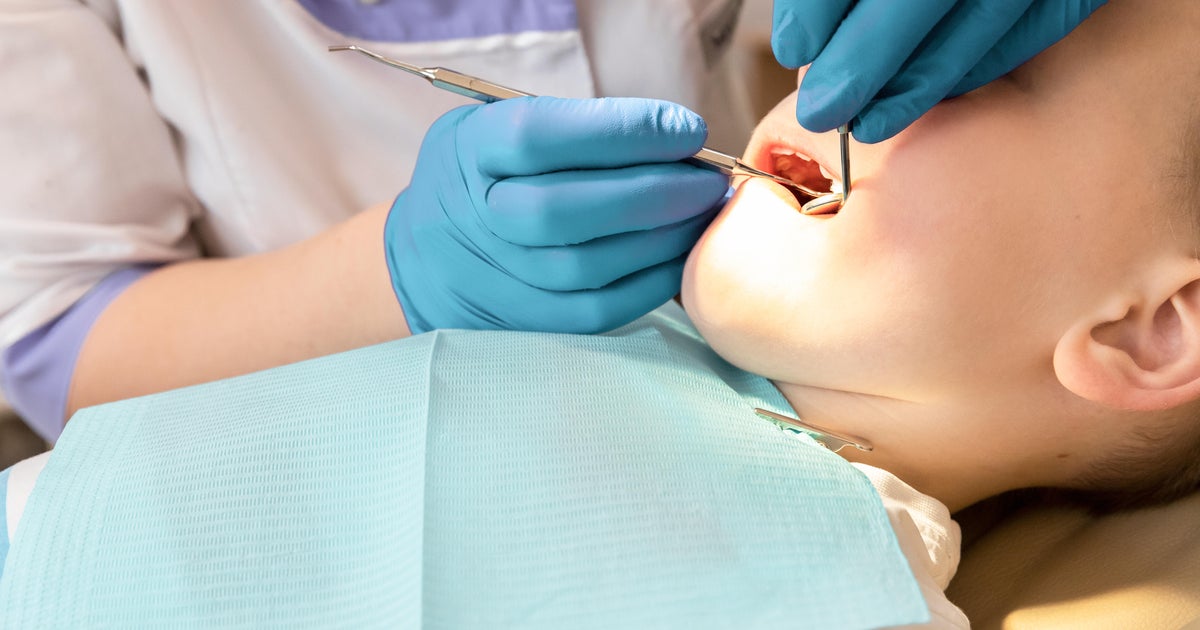
More kids are in ERs for tooth pain. Budget cuts and fluoride fights aren't helping.
Eight-year-old Jonah woke up one May morning with a swollen face and a toothache. He refused the pain medication that his mom, Geneva Reynolds, tried to give him. He didn't sleep or eat and cried constantly. Within a few days, Reynolds became so …
Source: CBS News - Neutral -
Published on Mar 8, 2026

Menstrual products contain toxic chemicals, shows research
About half of the global population menstruates at some point in their lives. Disposable products, such as tampons and pads, are some of the most popular products used around the globe to manage menstrual flow. Unfortunately, studies have shown that many …
Source: Scroll.in - Center-left -
Published on Mar 4, 2026

FDA Issues Revised Cybersecurity Premarket Submission Guidance To Align With Quality Management System Regulation
The United States Food and Drug Administration (FDA) has released updated cybersecurity guidance titled Cybersecurity in Medical Devices: Quality System Considerations and Content of Premarket Submissions – Guidance for Industry and Food and Drug …
Source: JD Supra - California - Neutral -
Published on Mar 3, 2026

FDA recalls Great Value Cottage Cheese sold in 24 states, including Illinois
CHAMPAIGN COUNTY (Chambana Today) — Consumers are urged to check their refrigerators after the U.S. Food and Drug Administration announced a recall of several Great Value cottage cheese products sold at Walmart stores in 24 states — including Illinois. The …
Source: WIXY 100.3 - Illinois - Pending -
Published on Mar 8, 2026

The great American vape patchwork (and what it says about us)
Contrary to common belief, there is no federal ban on disposable vapes in the United States. There is also no federal anything else on disposable vapes in the United States, which is how we ended up with the current situation: a patchwork of state laws so …
Source: Caribbean National Weekly - Neutral -
Published on Mar 8, 2026

The missing pieces of menopause science
When Rachel Pope started studying the menopause transition a decade ago, she hadn’t lived it herself. So, she asked her three older sisters who they would turn to for help if they experienced the sleep problems, thinning hair, low libido and hot flushes …
Source: Nature - Neutral -
Published on Mar 3, 2026

Lafayette father first in Colorado to receive newly approved pancreatic cancer drug
A Lafayette father's battle with stage 4 pancreatic cancer is taking a hopeful turn as he becomes the first in Colorado to receive a newly approved targeted therapy. DENVER — A Lafayette father with stage 4 pancreatic cancer is believed to be the first …
Source: KUSA 9 - Colorado - Neutral -
Published on Mar 2, 2026

'Quest' cat food recall expanded as company halts all sales over vitamin deficiency risk
A Utah pet food company has halted sales of its entire Quest product line and expanded a voluntary recall to include two lots of frozen cat food after disclosing potentially low thiamine levels that the company says can cause thiamine deficiency and …
Source: The Washington Times - Washington D.C. - Right-wing -
Published on Mar 3, 2026

FDA Accepts Supplemental NDA to Expand Phentolamine Ophthalmic Solution Use to Presbyopia
The FDA has accepted a supplemental new drug application (sNDA) seeking to expand the indication for Phentolamine Ophthalmic Solution to include the treatment of presbyopia, according to press releases from Opus Genetics and Viatris. Ryzumvi was originally …
Source: Medical Dialogues - Indeterminate -
Published on Mar 2, 2026

Business Buzz: Denver’s AI prenatal breakthrough and Colorado’s return to the moon
Colorado is pioneering future industries, from FDA-approved AI in prenatal care to crafting the Orion spacecraft for lunar missions. COLORADO, USA — Two Colorado stories are commanding national attention this week. One is transforming prenatal medicine …
Source: KUSA 9 - Colorado - Neutral -
Published on Mar 2, 2026

FDA Okays New Indications for Once-Weekly Growth Hormone
The US FDA has approved Novo Nordisk’s once-weekly growth hormone somapacitan-beco (Sogroya) for three new pediatric indications. The injectable long-acting growth hormone, in doses of 5 mg, 10 mg, and 15 mg, is now indicated for children aged 2.5 years …
Source: Medscape - Neutral -
Published on Feb 27, 2026

FDA cautions consumers about cottage cheese recall
Multiple cottage cheese products sold at Walmart under the Great Value brand are being recalled due to a possible health risk, the U.S. Food and Drug Administration reports. Announced by the FDA Wednesday, Feb. 25, the Great Value (GV) recall includes …
Source: Oelwein Daily Register - Iowa - Pending -
Published on Mar 1, 2026

Colorado family pushes for change after rare disease clinical trial abruptly ends
This week marks Rare Disease Week, a time when families across the country are sharing their struggles with access to treatments and clinical trials, and their hopes for change, with lawmakers and federal health officials. A Colorado family is now adding …
Source: CBS 4 - Colorado - Neutral -
Published on Feb 27, 2026

Gwyneth Paltrow looks chic in a winter white coat and skirt as she raises a glass at boozy pop-up launch in Colorado - ...
Gwyneth Paltrow enjoyed a tipple on Thursday after travelling to Colorado for a new VIP pop-up launch, hosted by Californian wine brand Meiomi Pinot Noir. The Oscar-winning actress looked appropriately stylish in a sweeping white frock coat and matching …
Source: Daily Mail - Right-wing -
Published on Feb 27, 2026

Food recall includes cottage cheese sold at Mississippi…
Feb. 26, 2026, 1:58 p.m. CT If you bought cottage cheese recently, check your refrigerator. Great Value brand products sold at Walmart grocery stores in Mississippi and 23 other states are part of a food recall. According to the U.S. Food and Drug …
Source: Hattiesburg American - Mississippi - Pending
Featured Releases
-
Published on March 9, 2026 - 07:05 GMT
Top Natural Sweeteners Manufacturers Continue to Gain Ground as the Global Market Shifts Toward Healthier Ingredients
CHENGDU CITY, CHINA, March 9, 2026 /EINPresswire.com/ -- The global food and beverage industry is undergoing a significant transformation. Consumers in markets across North America, Europe, and Asia-Pacific are actively reducing their intake of refined …
Distribution channels: Beauty & Hair Care, Chemical Industry ... -
Published on March 7, 2026 - 17:00 GMT

Robooter Launches U.S. Direct to Consumer Website, Bringing FDA Cleared Smart Power Wheelchairs to U.S. Customers
Robooter.us goes live with two flagship models and a roadmap that includes advanced assisted driving features targeted for Q4 2026 WILMINGTON, DE, UNITED STATES, March 7, 2026 /EINPresswire.com/ -- Robooter, a global mobility technology company with …
Distribution channels: Healthcare & Pharmaceuticals Industry ...
-
Published on March 9, 2026 - 06:53 GMT

Japan Contract Sterile Fill-Finish Services Market Growth Projections US$398.28Mn by 2033 | Growth Biologics & Vaccines
Japan Contract Sterile Fill-Finish Services Market Expands as Biologics Manufacturing and Regulatory Support Accelerate Outsourcing Demand TOKYO, OSAKA, JAPAN, March 9, 2026 /EINPresswire.com/ -- Market Size and Growth 2026: The rapid rise …
Distribution channels: Healthcare & Pharmaceuticals Industry ... -
Published on March 8, 2026 - 20:49 GMT

Alternative to Meds Center Highlights Long-Term Fanapt Risks and Individualized Antipsychotic Tapering Support
Sedona inpatient program educates on iloperidone side effects, anticholinergic burden, and holistic alternatives for schizophrenia and bipolar symptoms SEDONA, AZ, UNITED STATES, March 8, 2026 /EINPresswire.com/ -- Alternative to Meds Center ( …
Distribution channels: Agriculture, Farming & Forestry Industry, Consumer Goods ...
-
Published on March 8, 2026 - 20:40 GMT

Alternative to Meds Center Highlights Clinical Approach to Safe Zoloft Tapering and SSRI Withdrawal Support
Arizona-based inpatient program provides medically supervised sertraline tapering with holistic neuro-chemical restoration strategies SEDONA, AZ, UNITED STATES, March 8, 2026 /EINPresswire.com/ -- Alternative to Meds Center (ATMC) is raising …
Distribution channels: Agriculture, Farming & Forestry Industry, Culture, Society & Lifestyle ... -
Published on March 8, 2026 - 20:44 GMT

Alternative to Meds Center Highlights Prozac Risks, SSRI Controversy, & Safer Paths for Fluoxetine Tapering and Recovery
Sedona inpatient program emphasizes due diligence, withdrawal-aware care, and holistic alternatives for depression and anxiety support SEDONA, AZ, UNITED STATES, March 8, 2026 /EINPresswire.com/ -- Alternative to Meds Center (ATMC) is increasing …
Distribution channels: Agriculture, Farming & Forestry Industry, Culture, Society & Lifestyle ... -
Published on March 3, 2026 - 19:39 GMT

FDA Warns 30 Telehealth Companies Against Illegal Marketing of Compounded GLP-1s
For Immediate Release: March 03, 2026 The U.S. Food and Drug Administration today announced the issuance of 30 warning letters to telehealth companies for making false or misleading claims regarding compounded GLP-1 products offered on their websites. “It …
Distribution channels: Food & Beverage Industry -
Published on March 3, 2026 - 20:01 GMT

Sanaregen™ Vision Therapeutics Receives FDA Clearance for Clinical Trial to Treat Retinal Degeneration
NASHVILLE, TN, UNITED STATES, March 3, 2026 /EINPresswire.com/ -- Sanaregen Vision Therapeutics, a clinical-stage regenerative medicine company, today announced clearance by the U.S. Food and Drug Administration to conduct a Phase I/II clinical trial for …
Distribution channels: Banking, Finance & Investment Industry, Healthcare & Pharmaceuticals Industry ...
Online News Monitoring Made Simple
Established in 1995, EIN Newsdesk helps millions of users track breaking news across thousands of trusted websites. Users may set up custom email newsletters and RSS feeds or search among thousands of preset news sections. As a member, you may also submit your own news using the EIN Presswire distribution service. Membership is free and we do not sell or lease any information about you. Press release distribution, dedicated APIs, and custom services generate the revenue that allow us to offer you free basic access.
News by Country & by Industry
Track news across more than 5,000 sources by country or industry or search our real-time news index.
News Alerts & News Feeds
News page results can be outputted as RSS or received daily by email. You may also use our NewsPlugin for WordPress.
Submit News
Our EIN Presswire's distribution footprint reaches millions. You can upload news which is re-published through EIN Newsdesk, fed into Twitter and Facebook, and onto partner sites, Google News, Bing News, etc.
World Media Directory
This is a listing of the world’s TV stations, radio stations, newspapers, and blogs. Last update: 02/12/2026

Food & Beverage Industry Press Releases
-
Published on March 9, 2026 - 11:28 GMT

Vegetable Oil Market to Expand Through 2034 Fueled by Growing Population and Increasing Food Industry Use
IMARC Group’s latest report highlights steady market growth driven by changing dietary habits, rising industrial demand, and advancements in the industry. ST. BROOKLYN, NY, UNITED STATES, March 9, 2026 /EINPresswire.com/ -- The global Vegetable …
Distribution channels: Consumer Goods, Food & Beverage Industry ... -
Published on March 9, 2026 - 10:56 GMT

Golden Waffle Launches UK-Blended Waffle Mix to Help Hospitality Operators Cut Costs
Golden Waffle introduces a UK-blended just-add-water waffle mix designed to reduce kitchen prep, improve consistency and deliver stronger profit margins. MANCHESTER , LANCASHIRE, UNITED KINGDOM, March 9, 2026 /EINPresswire.com/ -- Manchester, …
Distribution channels: Companies, Consumer Goods ... -
Published on March 9, 2026 - 10:53 GMT

Millet Market to Reach USD 18.72 Billion by 2032 at 4.89% CAGR: Rising Demand for Nutritious Grains
The global millet market was valued at USD 13.40 billion in 2025 and is projected to reach USD 18.72 billion by 2032, growing at a CAGR of 4.89%. ROCKVILLE , MD, UNITED STATES, March 9, 2026 /EINPresswire.com/ -- The Global Millet Market is undergoing …
Distribution channels: Food & Beverage Industry ... -
Published on March 9, 2026 - 10:25 GMT

THE EU FAB 6 SHINES A LIGHT ON THE 'GI ECONOMY'
New insights into the economic value of geographical indication products and how consumers can recognise them ROMA, ITALY, March 9, 2026 /EINPresswire.com/ -- New figures confirm the growing economic impact of Europe’s geographical indication products. …
Distribution channels: Agriculture, Farming & Forestry Industry, Business & Economy ... -
Published on March 9, 2026 - 10:21 GMT

THE EU FAB 6 PUNTA I RIFLETTORI SULLA 'GI ECONOMY'
Nuovi dati sul valore economico dei prodotti a indicazione geografica e su come i consumatori possono riconoscerli ROMA, ITALY, March 9, 2026 /EINPresswire.com/ -- German and French versions below. Nuovi dati confermano il crescente impatto …
Distribution channels: Agriculture, Farming & Forestry Industry, Business & Economy ... -
Published on March 9, 2026 - 10:10 GMT

Cutting Energy Costs and Extending Vacuum Pump Lifetime in Food Packaging
To optimize performance and reduce energy consumption, Kepak relied on Busch Vacuum Solutions, a partner they have worked with for around 15 years now. ABERDEEN, SCOTLAND, March 9, 2026 /EINPresswire.com/ -- When a family sets a premium cut of beef on …
Distribution channels: Companies, Food & Beverage Industry ... -
Published on March 9, 2026 - 10:00 GMT

BoxLogix Delivers Automated Sortation System for Regional Healthcare Distribution Hub
Automation streamlines parcel receiving and routing for a regional healthcare network, improving efficiency and ensuring accurate distribution to hospitals. PRAIRIE DU CHIEN, WI, UNITED STATES, March 9, 2026 /EINPresswire.com/ -- BoxLogix, a …
Distribution channels: Automotive Industry, Food & Beverage Industry ... -
Published on March 9, 2026 - 09:00 GMT

ABA, RBA & Baking Association of Canada sign MOU to forge strategic partnership
ABA, RBA & BAC sign MOU to advance North American baking through shared advocacy, resources & programming while remaining independently managed. OAKVILLE, ON, CANADA, March 9, 2026 /EINPresswire.com/ -- Today, the American Bakers Association ( …
Distribution channels: Business & Economy, Conferences & Trade Fairs ... -
Published on March 9, 2026 - 08:30 GMT
AI Startup TUBR Selected for International Accelerator While Running Live Trials in Phoenix, Arizona
Sheffield-based TUBR—now co-located in Arizona—selected for Andorra Open Valley while demand forecasting platform goes live with Phoenix hospitality businesses PHOENIX, AZ, UNITED STATES, March 9, 2026 /EINPresswire.com/ -- TUBR, a UK-based AI …
Distribution channels: Business & Economy, Food & Beverage Industry ... -
Published on March 9, 2026 - 07:38 GMT

Food Grade Ethanol Market Witness Remarkable Growth, Projected to Reach US$4.7 Bn by 2033 - Persistence Market Research
North America is expected to dominate with an estimated 32% of the food-grade ethanol market share in 2026, with the United States as the primary contributor LONDON, LONDON, UNITED KINGDOM, March 9, 2026 /EINPresswire.com/ -- The global food …
Distribution channels: Food & Beverage Industry ... -
Published on March 9, 2026 - 07:05 GMT
Top Natural Sweeteners Manufacturers Continue to Gain Ground as the Global Market Shifts Toward Healthier Ingredients
CHENGDU CITY, CHINA, March 9, 2026 /EINPresswire.com/ -- The global food and beverage industry is undergoing a significant transformation. Consumers in markets across North America, Europe, and Asia-Pacific are actively reducing their intake of refined …
Distribution channels: Beauty & Hair Care, Chemical Industry ... -
Published on March 9, 2026 - 00:00 GMT

Personalized Nutrition & Supplements Market 2026:Leverage Customized Nutrition Solutions Accelerating Precision Wellness
The Business Research Company's Personalized Nutrition And Supplements Market Report 2026 – Market Size, Trends, And Global Forecast 2026-2035 LONDON, GREATER LONDON, UNITED KINGDOM, March 9, 2026 /EINPresswire.com/ -- "Personalized …
Distribution channels: Business & Economy, Food & Beverage Industry ... -
Published on March 7, 2026 - 19:15 GMT

Malatya Apricot | Dried Fruits – Nuts Supplier : Dates, Prunes, Raisin Delivery Worldwide with Quality, Reliability
Malatya Apricot | Dried Fruits – Nuts Supplier : Premium Dates, Prunes, Raisin and Nut Varieties Delivered Worldwide with Quality, Reliability, and Trust MALATYA, BATTALGAZI, TURKEY, March 8, 2026 /EINPresswire.com/ -- In an increasingly health …
Distribution channels: Agriculture, Farming & Forestry Industry, Companies ... -
Published on March 6, 2026 - 20:57 GMT

Confience to Exhibit at Pittcon 2026, Showcasing myLIMS and Headlining Key Presentations
Lab technology innovator brings its flagship LIMS platform and expert-led sessions to the world's premier science conference, March 9–11 in San Antonio, Texas. RALEIGH, NC, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- Confience, a leading …
Distribution channels: Chemical Industry, Energy Industry ... -
Published on March 6, 2026 - 20:53 GMT

This Girl Walks Into a Bar Announces Return of Bartending Services After One- Year Hiatus Following Palisades Fire
The beloved Los Angeles bartending team returns to the bar. LOS ANGELES, CA, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- After a year-long pause following the devastating Palisades Fire, This Girl Walks Into a Bar is proud to announce …
Distribution channels: Business & Economy, Culture, Society & Lifestyle ... -
Published on March 6, 2026 - 19:00 GMT

Authentic Kobe Beef experience lands in Kabukicho, Shinjuku, at Wagyu Tokku Japanese Steakhouse
Aimed at fulfilling travelers’ wish to try authentic Kobe Beef, in Japanese steak style in the heart of Tokyo. ADACHI-KU, TOKYO, JAPAN, March 6, 2026 /EINPresswire.com/ -- Heijoen Co., Ltd. (Head office: Adachi-ku, Tokyo; President and CEO: …
Distribution channels: Culture, Society & Lifestyle, Food & Beverage Industry ... -
Published on March 6, 2026 - 17:18 GMT

Pegasus Platinum Portable Truck Mount Hard-Surface Extractor Delivers Highest Power, 6,000 Hour Runtime
Truck Mount Power, Extended Distance Suction, and Portability Delivers Continuous-Duty Operations and and Increased Revenue to Professional Cleaning Crews VANCOUVER, WA, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- U.S. Products, the …
Distribution channels: Business & Economy, Education ... -
Published on March 6, 2026 - 15:00 GMT

Great Coffee Starts with Better Water: Bluewater Shows How at New York Coffee Fest 2026
Bluewater SuperiorOsmosis clears the slate, while LiquidRock rebuilds the minerals. Taste the difference March 8–10 at the Javits Center. NEW YORK, NY, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- Ask any barista what matters in a great cup, and …
Distribution channels: Companies, Culture, Society & Lifestyle ... -
Published on March 6, 2026 - 15:00 GMT

SquarePet to Showcase Veterinarian-Formulated Nutrition at the 2026 Global Pet Expo Booth #4838
AUSTIN, TX 78717, TX, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- SquarePet Nutrition, a leading U.S. manufacturer of premium pet nutrition products, will exhibit at the Global Pet Expo in Orlando, Florida. Attendees are invited to visit …
Distribution channels: Companies, Conferences & Trade Fairs ... -
Published on March 6, 2026 - 14:39 GMT

Chef Russell Jackson Announces Spring 2026 Public Programming and Culinary Venture Updates in New York
New York chef expands public-facing work across experiential dining and hospitality ventures as he appears in a nationally broadcast culinary competition NEW YORK, NY, UNITED STATES, March 6, 2026 /EINPresswire.com/ -- Chef Russell Jackson announced a …
Distribution channels: Culture, Society & Lifestyle, Food & Beverage Industry ...
Food & Beverages Events
No event was found.

- Track news from thousands of sources
- Submit news sources for inclusion in our news tracking algorithm
- Build your own customized email newsletters
- Use of our RSS Feeds
- Includes a complimentary EIN Presswire account plus 1-free release
- Uncluttered & advertising free news pages

