Native Orthopaedics Receives FDA 510k clearance for Dragonfly™ knotless quad core technology for Meniscal Root Repair
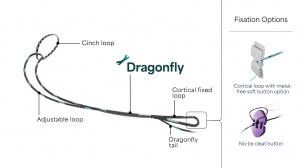
Dragonfly™ is a novel advanced quad core knotless finger trap technology that provides sports medicine surgeons instant retensioning capabilities for use in knee repair procedures. Users can easily create precise fixation in one fluid motion by simply pulling the end of the device, which tensions and locks, and can be retensioned as desired.
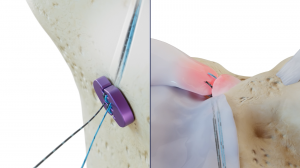
The Native Root System consists of two single-use sterile-packed Dragonfly devices, specifically designed to reapproximate a torn meniscal root back to the native footprint. The Dragonfly platform utilizes a patented design and manufacturing technique that offers greater mechanical strength compared to basic finger traps and other decades-old technologies.1 This “trap within a trap” architecture does not rely on a secondary locking feature to prevent slippage. The suture tension is transmitted directly to the surgeon’s fingertips, allowing improved control and feel during tensioning. Furthermore, the Dragonfly platform is designed to be retensionable, which may allow the surgeon to recover the “unrecoverable” creep.
“With the introduction of the Dragonfly platform to the market, Native Orthopaedics aims to improve the standard of care for all of sports medicine, starting with the meniscal root repair use case,” said Tom Gamache, CEO of Native Orthopaedics. “We are pleased to receive FDA-clearance for our breakthrough technology, which we believe will help significantly improve outcomes for these repairs. Demonstrating our commitment to rapid innovation in the sports medicine space, I am proud of our team for translating this from a concept to a clinically ready best-in-class solution in under a year. Also, our entire team is energized to bring this Dragonfly platform technology into additional opportunities in the future. Native's Dragonfly platform technology enables scalability of knotless across the continuum of sports medicine, while addressing the systemic limitations with current knotless solutions.”
Meniscal root repair is one of the fastest-growing procedures performed in orthopaedics worldwide, with an estimated underreported 200,000 procedures performed in the United States alone. The growth of this segment is driven by both patient demographics and a shift in activity levels of aging populations. Meniscal root repair is ripe for disruption due to the ongoing limitations of current surgical solutions:
• 78% of total knee replacements have a medial meniscus root tear at the time of surgery, indicating a potentially massively underserved market.2
• With current technology, nearly 40% of meniscal root repairs fail (2 in 5 repairs).3
• Clinically, just 3 mm of tissue creep leads to repair failure.4
• Further, just a 5 mm deviation of the fixation from the native attachment results in failure.5
Testing of the Dragonfly system has demonstrated significant advantages over the current standard of care and legacy products. Highlights from the biomechanical testing and simulated clinical use include the following:
• 290% increase in ultimate strength compared to market leader.1
• Less than 0.4 mm elongation under cyclic testing.1
“Our mission is patient-centered and surgeon-focused, and I believe the Dragonfly technology will empower surgeons to restore the native anatomy in ways that have previously not been allowed,” said Christian Anderson, MD, Founding Surgeon and Chief Medical Officer of Native Orthopaedics. “Meniscal root tears have been called a ‘silent epidemic’, and if managed conservatively, have dire consequences, including a 95% clinical failure rate with 50% conversion to knee arthroplasty within 10 years after the injury.6 Current repair techniques have unacceptably high failure rates and having evaluated all commercially available products for meniscal root repair, I can confidently say that the Native Root System is the simplest, most efficient, and best solution available to improve outcomes, all with no knots and no anchors.”
The Native Root System has been perfected through years of surgeon-driven, painstaking research and design advancements. The supporting instrumentation has been designed with the surgeon's input at every point along the way. The combined system specifically addresses the challenges facing current root systems regarding precise tensioning, low-creep fixation, tunnel accuracy, and suture passing misfires. The Native Root System will be available for commercial use in select locations starting in May 2025.
##
About the Native Root System powered by Dragonfly™®
Native Orthopaedics Root System is intended to be used for suture (soft tissue) fixation to bone in the knee for meniscal root repair. Dragonfly technology is protected by 12 issued patents, with 30+ additional patents under review including worldwide coverage.
About Native Orthopaedics
Native Orthopaedics is a privately held company actively fundraising pre-series A with a series A round anticipated in the coming months. Native Orthopaedics is committed to delivering innovative patient centric solutions to transform the sports medicine industry and make surgeries simpler and more reliable for health care professionals. Native Orthopaedics is a company built by a Surgeon for Surgeons. We are innovators and doers who want to help those with knee pain live life to its fullest. We’re committed to crafting creative, end-to-end surgical solutions and innovative tools that push the field of orthopaedics into the future with simpler, more effective solutions that preserve our native anatomy.
Native Orthopaedics has one commercial-stage solution, the Native Root System powered by Dragonfly™, a novel, next-generation, knotless suture technology – designed to enhance repair biomechanics and reduce clinical failure with its novel quad core trap within a trap architecture. The company also has one end-stage development product targeting ACL repairs, among other indications, and a robust pipeline with anticipated future product launches within the knee, shoulder, and ankle. To learn more about Native Orthopaedics, visit www.NativeOrtho.com.
Investor inquiries can be directed to tgamache@nativeortho.com
Citations:
1. Data on file with Native Orthopaedics, Inc.
2. Choi ES, et al. KSRR. 2015
3. Feucht MJ, et al. Arthroscopy. 2015
4. Steinman DB, et al. Arthroscopy. 2019
5. LaPrade CM, et al. Am J Sports Med. 2015
6. Krych AJ, et al. Am J Sports Med. 2023
Travis Small
Native Orthopaedics
+1 617-538-9041
email us here
Visit us on social media:
LinkedIn
Distribution channels: Healthcare & Pharmaceuticals Industry, Technology
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Submit your press release